Caplin Point Laboratories announced that the company's state of art injectable plant near Gummidipoondi, Tamil Nadu to cater regulated markets USA, EU, Brazil, South Africa etc., has commenced its commercial production on Mar. 27, 2014. This will now enable export of these products to semi regulated and non regulated markets.
 The company has now initiated the process of submitting required documents for obtaining approval from regulatory bodies such as UKMHRA, ANVISA, BRAZIL, USFDA, EUGMP, which would enable export of injectables in various dosage forms to the regulated markets besides offering contract manufacturing for export to these markets.
The company has now initiated the process of submitting required documents for obtaining approval from regulatory bodies such as UKMHRA, ANVISA, BRAZIL, USFDA, EUGMP, which would enable export of injectables in various dosage forms to the regulated markets besides offering contract manufacturing for export to these markets.
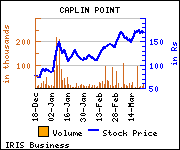
Shares of the company gained Rs 8.35, or 5%, to trade at Rs 175.50. The total volume of shares traded was 27,419 at the BSE (11.22 a.m., Thursday).